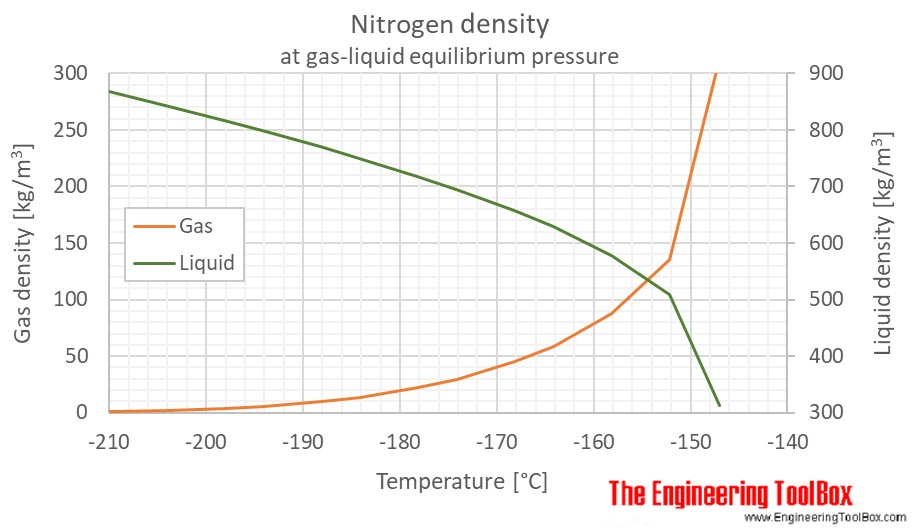
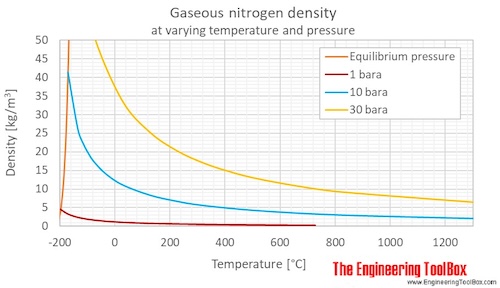
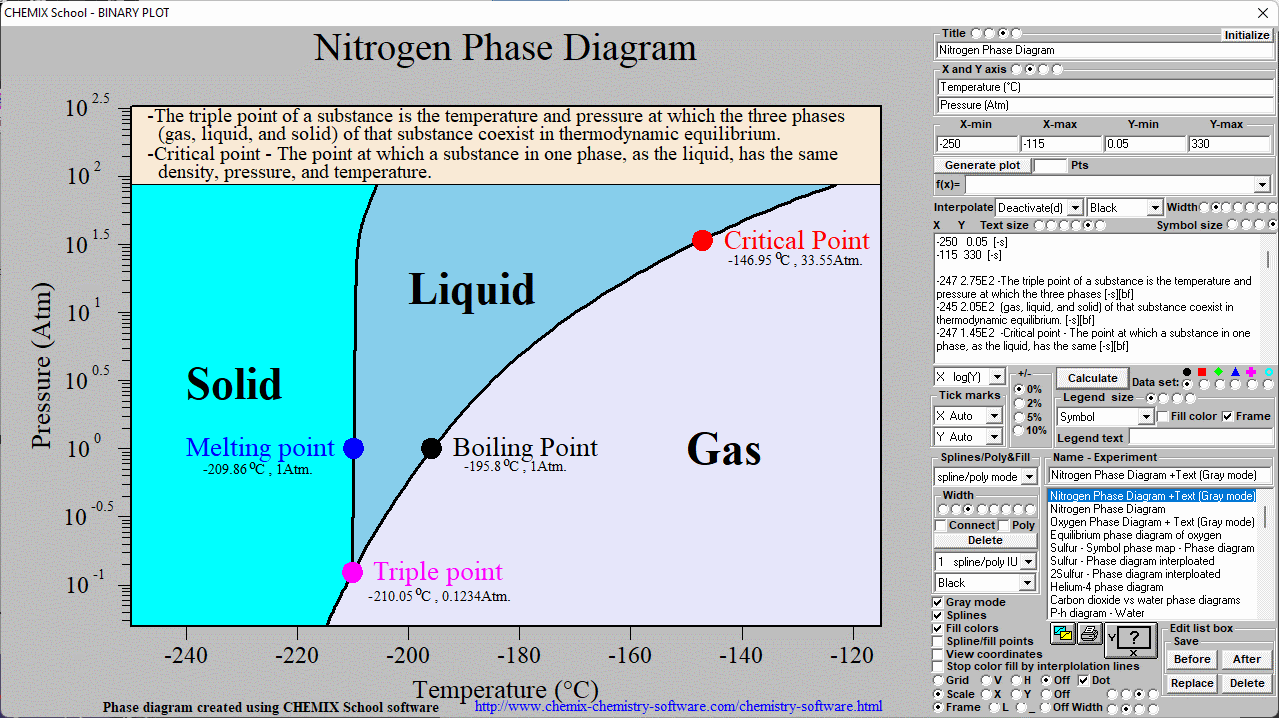
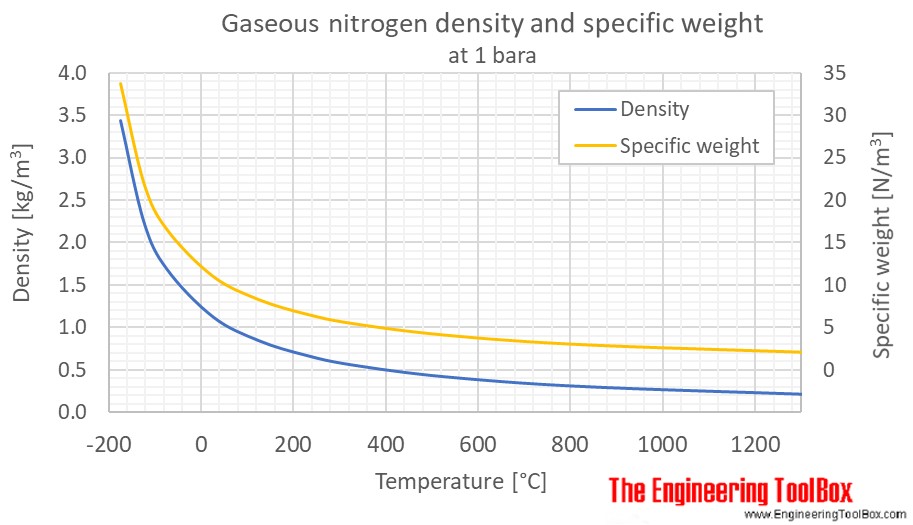
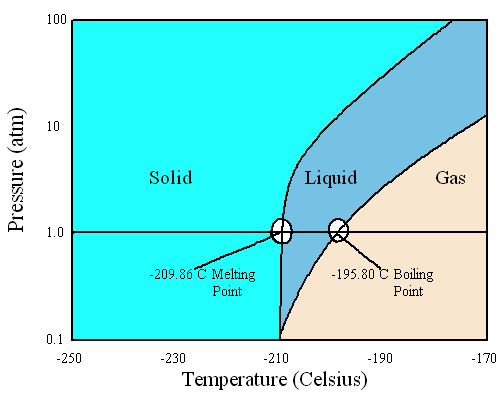
SOLVED: Yueslion 4 ( 1 pointi A sample of nitrogen gas is produced in a reaction and collected under water in a graduated cylinder: The temperature is 26.0 %C and the volume

A 15 g mass of nitrogen gas is enclosed in ayessel at a temperature 27 ^ ∘C . Amount of heat transferred to the gas, so that rinsvelocity of molecules is doubled,

Electrical conductivity of nitrogen as a function of temperature (ρ =... | Download Scientific Diagram
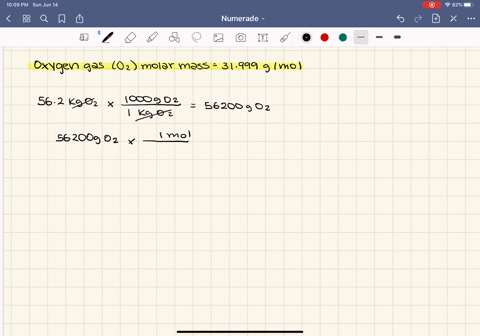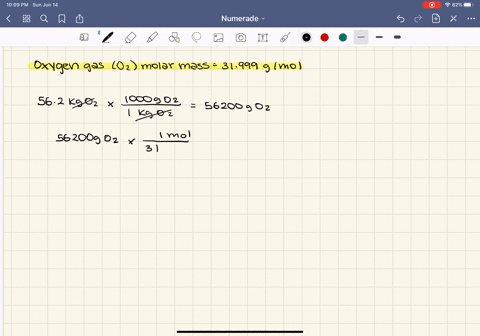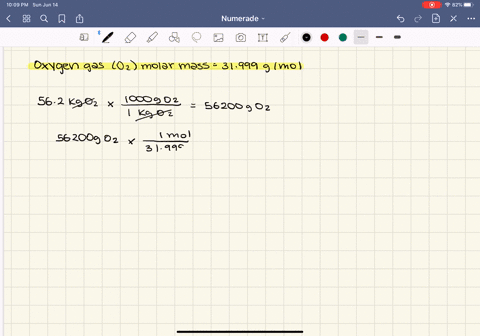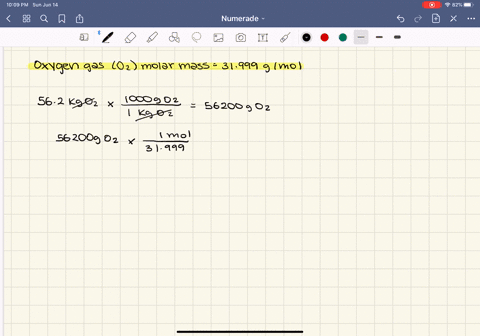
SOLVED:At what temperature does 16.3 g of nitrogen gas have a pressure of 1.25 atm in a 25.0-L tank?

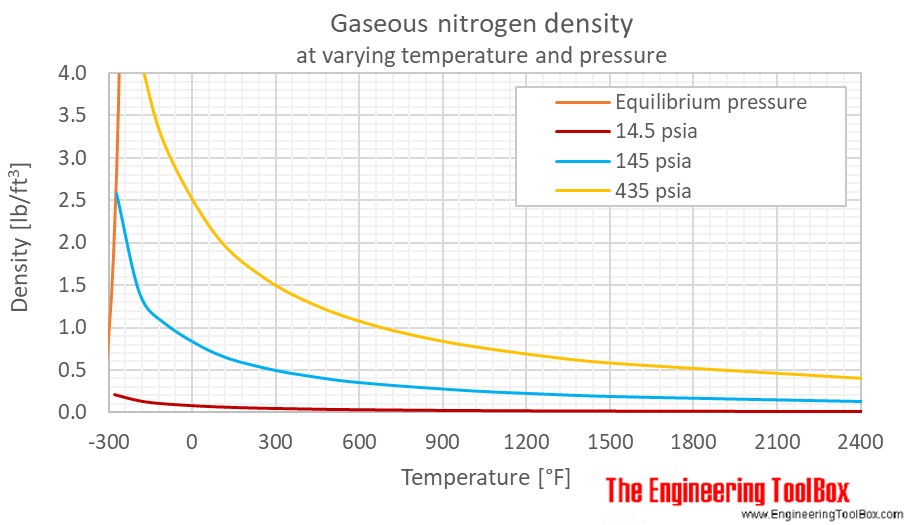
SOLVED: Density Practice Calculate the density of nitrogen gas at 125 degrees Cand 755 mmHg: Given: N1 T1J59€ +273= 3G8k P+7S5mnkx4= 0.9 G3661 7bd+bWf Needed:D= m Plan: AsSmc md Nj SG) -
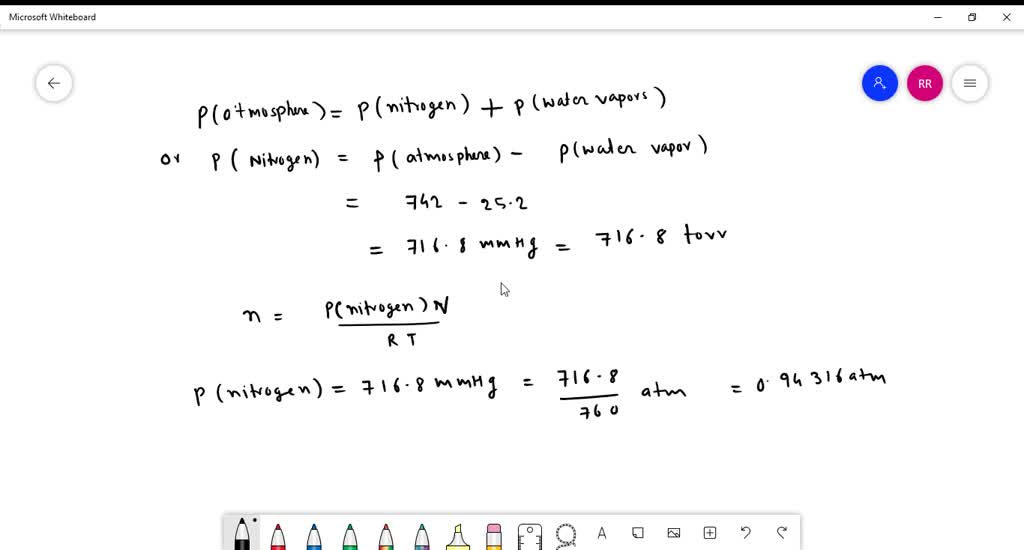
SOLVED: Question 1 (1 point) A sample of nitrogen gas is produced in a reaction and collected under water in a graduated cylinder. The temperature is 26.0 °C and the volume of